|
1/10/2024 0 Comments Cathode negative or positive
If the former takes electrons and the latter provides them?Ĭhemists worry about the direction of current flow to determine cathodeness,īut in electronics anode and cathode are fixed properties of the device. 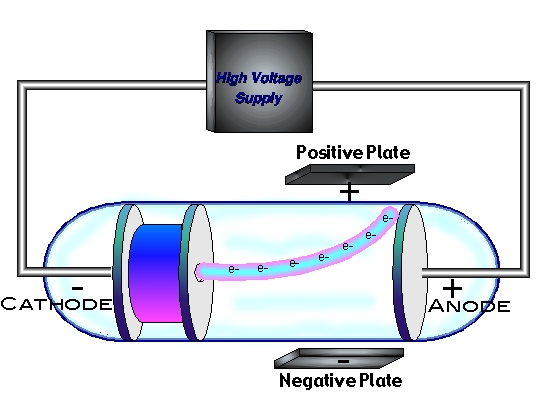
the battery cell), whereas cathode is the positive terminal since that is where the current flows out of the device. The anions, negative ions, flow toward the anode to balance the positive charge of the cations that are released from the electrode. in a galvanic cell, anode is the negative terminal because it is where the current flows into 'the device' (i.e. Why do we say Cathode is negative and Anode is positive The cations, positive ions, flow toward the cathode to replace the cations that are being picked up at the electrode. if the cell is being charged or discharged) which terminal will be anode and which cathode not it electrical potential. it depends on the direction of the current (ie. The voltage on the anode is immaterial, so I can't say if it's attracting or repelling electrons. The anode oxidises releasing electrons which flow into the external circuitĮlectrons have negative charge so the electrons flowing out of the anode mean conventional current flowing into it. It's called an anode because that's the terminology chemists chose. The cathode is where reduction takes place (the cathode gains electrons).īut I am confused why is the anode called anode if it is providing electrons instead of attracting them? I know that in a battery: The anode is where oxidation takes place (the anode losses electrons). In chemistry the anode is the terminal that is producing external electron current and the cathode is consuming an equal current. It's not usual to talk about anode and cathode of a battery (or cell) in electroncs, it's more a chemistry thing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
